On the other hand, if we throw an ice cube across the room, all of its constituent water molecules fly across the room together. To compress the solid, the molecules must be pushed still closer together, which requires inordinate force. We attribute the incompressibility of a solid to repulsive forces between its constituent molecules they have come so close to one another that repulsive forces between them have become important. Now, two distinguishing features of a solid are that it retains its shape and that it is almost incompressible. The crystal must be perfect, or else there will be some inherent disorder. 
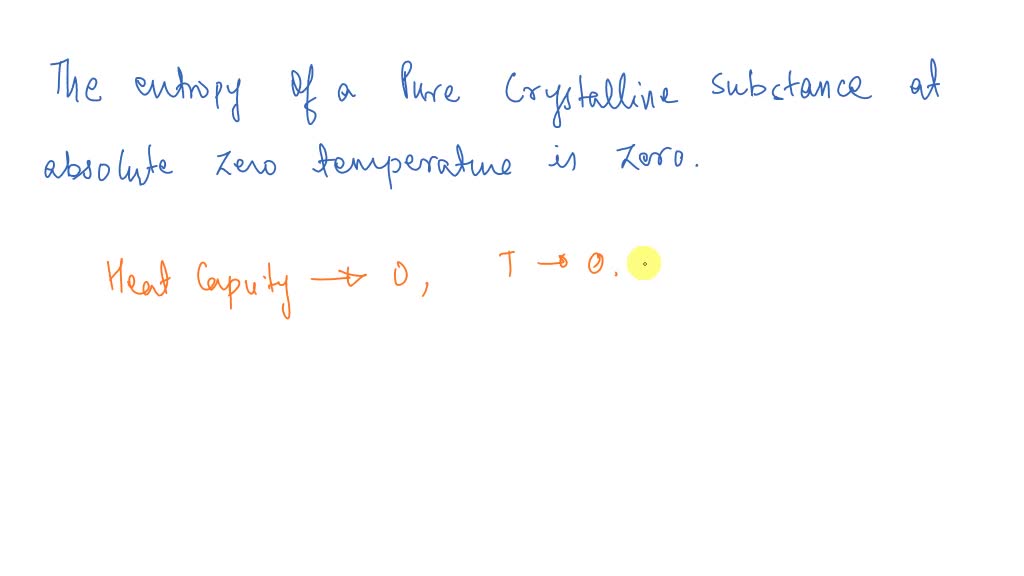
Third law: The entropy of a perfect crystal is zero when the temperature of the crystal is equal to absolute zero (0 K). If we cool the liquid further, it freezes to a solid. The third law of thermodynamics defines absolute zero on the entropy scale. To recognize that real gas molecules both attract and repel one another, we need only remember that any gas can be liquefied by reducing its temperature and increasing the pressure applied to it. Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License. Robinson, PhD, Richard Langley, Klaus Theopold We recommend using aĪuthors: Paul Flowers, William R. Use the information below to generate a citation. Then you must include on every digital page view the following attribution: The third law of thermodynamics provides an absolute reference point for the determination of entropy. If you are redistributing all or part of this book in a digital format, Then you must include on every physical page the following attribution: If you are redistributing all or part of this book in a print format, Want to cite, share, or modify this book? This book uses the This book may not be used in the training of large language models or otherwise be ingested into large language models or generative AI offerings without OpenStax's permission. Only a perfectly ordered, crystalline substance at absolute zero would exhibit no. 
Vibrational, rotational, and translational motions of a carbon dioxide molecule are illustrated here. A summary of these three relations is provided in Table 16.1. The entropy of any perfectly ordered, crystalline substance at absolute zero is zero. The third law derived from experiments looking at the behavior of heat capacities and entropy at lower and lower temperatures. The third law, like the other laws of thermodynamics, is derived from empirical observations made by scientists studying the behavior of thermodynamic systems. These results lead to a profound statement regarding the relation between entropy and spontaneity known as the second law of thermodynamics: all spontaneous changes cause an increase in the entropy of the universe. The Third Law and calculation of absolute entropies. This process involves no change in the entropy of the universe. 
In this case, the system and surroundings experience entropy changes that are equal in magnitude and therefore sum to yield a value of zero for Δ S univ. Over time, complex organisms evolved from much simpler ancestors, representing a large decrease in entropy of the Earth’s biosphere. The temperature difference between the objects is infinitesimally small, T sys ≈ T surr, and so the heat flow is thermodynamically reversible. Some people misunderstand the second law of thermodynamics, stated in terms of entropy, to say that the process of the evolution of life violates this law.This process involves a decrease in the entropy of the universe. The magnitude of the entropy change for the surroundings will again be greater than that for the system, but in this case, the signs of the heat changes will yield a negative value for Δ S univ. The arithmetic signs of q rev denote the gain of heat by the system and the loss of heat by the surroundings.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
